Insulin Pump & CGM Therapy for Children: The MiniMed™ 770G System
The Medtronic MiniMed™ 770G insulin pump system is the technology that adapts to you! Approved for ages 2 and up, this new, FDA-approved system automatically adjusts background insulin every 5 minutes.** By using real-time glucose readings from the integrated Guardian Connect continuous monitoring system, the system determines the amount of insulin needed based on your body’s needs.
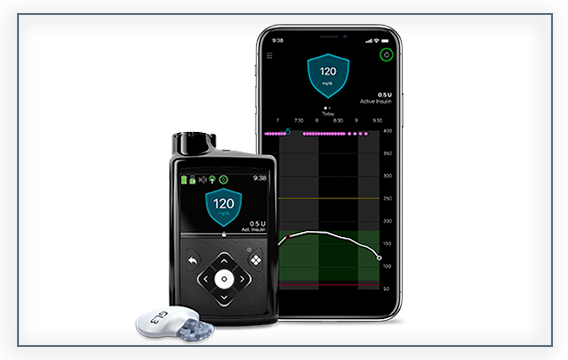
The Medtronic MiniMed™ 770G system combines insulin pump therapy and continuous glucose monitoring (CGM). The system is able to make adjustments to the amount of background insulin delivered based on your child’s needs, helping reduce highs and lows.*
This system also makes dinner time for your child incredibly easy! Enter the number of carbs that he/she will be eating, and the system will automatically calculate a mealtime insulin dose.
Multiple Features That Make Managing Your Child’s Diabetes Easier:
- Smartguard™ Auto Mode helps prevent glucose highs and lows
- MiniMed™ Mobile App allows your child to view his/her pump and CGM info on a smartphone
- Carelink™ Connect App brings more peace of mind for parents
Learn How The MiniMed™ 770G Insulin Pump System Can Help Your Child Prevent Glucose Highs And Lows!
Ready to get started with a MiniMed 770G insulin pump?
Apply Today* Refers to SmartGuard™ Auto Mode. Some user interaction required. Individual results may vary.
IMPORTANT SAFETY INFORMATION: MINIMED™ 770G SYSTEM WITH SMARTGUARD™ TECHNOLOGY
The MiniMed™ 770G system is intended for continuous delivery of basal insulin (at user selectable rates) and administration of insulin boluses (in user selectable amounts) for the management of type 1 diabetes mellitus in persons two years of age and older requiring insulin as well as for the continuous monitoring and trending of glucose levels in the fluid under the skin. The MiniMed™ 770G System includes SmartGuard™ technology, which can be programmed to automatically adjust delivery of basal insulin based on continuous glucose monitoring (CGM) sensor glucose values (SG) and can suspend delivery of insulin when the SG value falls below or is predicted to fall below predefined threshold values.
The Medtronic MiniMed™ 770G System consists of the following devices: MiniMed™ 770G Insulin Pump, the Guardian™ Link (3) Transmitter, the Guardian™ Sensor (3), one-press serter, the Accu-Chek® Guide Link blood glucose meter, and the Accu- Chek®Guide Test Strips. The system requires a prescription.
The Guardian™ Sensor (3) has not been evaluated and is not intended to be used directly for making therapy adjustments, but rather to provide an indication of when a fingerstick may be required. All therapy adjustments should be based on measurements obtained using a blood glucose meter and not on values provided by the Guardian™ Sensor (3).
All therapy adjustments should be based on measurements obtained using the Accu-Chek® Guide Link blood glucose meter and not on values provided by the Guardian™ Sensor (3). Always check the pump display to ensure the glucose result shown agrees with the glucose results shown on the Accu-Chek® Guide Link blood glucose meter. Do not calibrate your CGM device or calculate a bolus using a blood glucose meter result taken from an alternative site. It is not recommended to calibrate your CGM device when sensor or blood glucose values are changing rapidly, e.g., following a meal or physical exercise.
WARNING: Do not use the MiniMed™ 770G system until appropriate training has been received from a healthcare professional. Training is essential to ensure the safe use of the MiniMed™ 770G system.
Pump therapy is not recommended for people whose vision or hearing does not allow recognition of pump signals and alarms. Pump therapy is not recommended for people who are unwilling or unable to maintain contact with their healthcare professional. The safety of the MiniMed™ 770G system has not been studied in pregnant women. For complete details of the system, including product and important safety information such as indications, contraindications, warnings and precautions associated with system and its components, please consult http://www.medtronicdiabetes.com/important-safety- information#minimed-770g and the appropriate user guide at http://www.medtronicdiabetes.com/download-library.
Tags: Diabetes, Diabetes Management, Diabetes Supplies, Insulin Pump Therapy, Insulin Pumps, Medtronic



