FOOTNOTES
*Taking a bolus 15 – 20 minutes before a meal helps to keep blood sugar levels under control after eating.
†Refers to auto correct, which provides bolus assistance. Can deliver all auto correction doses automatically without user interaction, feature can be turned on and off.
§Refers to SmartGuard™ feature. Individual results may vary.
Δ Glucose targets should be set following a consultation with your healthcare provider.
Ω Fingersticks required in manual mode and to enter SmartGuard™. If symptoms don’t match alerts and readings, use a fingerstick. Refer to user guide. Pivotal trial participants spend avg of > 93% in SmartGuard™.
Φ Smart devices sold separately. For a list of compatible devices, visit http://bit.ly/CheckDevices.
MINIMED™ SYSTEM SOFTWARE UPGRADE PROGRAM
This program allows MiniMed™ 770G system users a software upgrade at no charge until April 27, 2024.
• Software upgrade process is subject to change. The process may result in a healthcare provider office visit and may result in out-of-pocket expenses such as co-pay, depending on insurance coverage. The process may require the completion of product related training.
• This program includes the insulin pump software only. CGM products and consumables are not included in this offer.
• Warranty dates do not reset. The replacement warranty date of an insulin pump upgraded with the software will remain the same as the replacement warranty date of the initial pump purchased. Your warranty date does not impact eligibility for a software update.
• MiniMed™ pumps and software have certain labeling restrictions that may exclude certain populations, including age and type of diabetes. We recommend you consult your healthcare provider regarding your eligibility to receive the software upgrade based on the labeling of the upgrade, which may differ from the labeling of the MiniMed™ 770G pump.
• Insurance coverage and plan design varies and may impact the ability to upgrade. Coverage is contingent upon your health insurance medical policy guidelines. Restrictions and medical necessity requirements may apply.
• A valid prescription will be required for this software upgrade. All participants should consult their healthcare provider for all therapy related decisions.
• Program terms and conditions are subject to change at any time and without notice. Other restrictions may apply.
IMPORTANT SAFETY INFORMATION
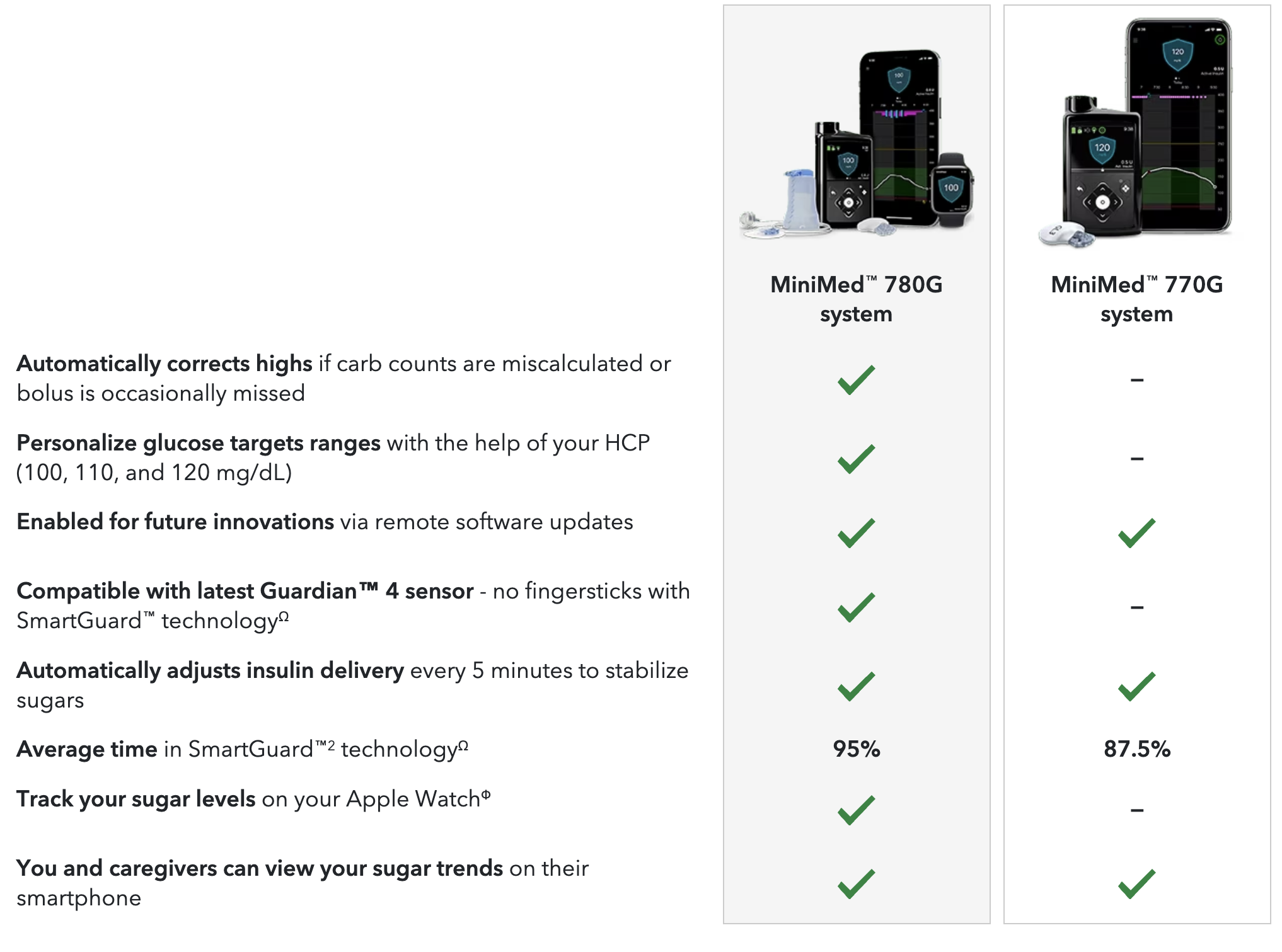
Medtronic MiniMed™ 780G System With SmartGuard™ Technology with Guardian™ 4 Sensor
The MiniMed™ 780G system is intended for continuous delivery of basal insulin at selectable rates, and the administration of insulin boluses at selectable amounts for the management of type 1 diabetes mellitus in persons seven years of age and older requiring insulin as well as for the continuous monitoring and trending of glucose levels in the fluid under the skin. The MiniMed™ 780G system includes SmartGuard™ technology, which can be programmed to automatically adjust insulin delivery based on the continuous glucose monitoring (CGM) sensor glucose values and can suspend delivery of insulin when the sensor glucose (SG) value falls below or is predicted to fall below predefined threshold values.
The Medtronic MiniMed™ 780G system consists of the following devices: MiniMed™ 780G insulin pump, the Guardian™ 4 transmitter, the Guardian™ 4 sensor, One press serter, the Accu-Chek™ Guide Link blood glucose meter, and the Accu-Chek™ Guide test strips. The system requires a prescription from a healthcare professional.
The Guardian™ 4 sensor is intended for use with the MiniMed™ 780G system and the Guardian 4 transmitter to monitor glucose levels for the management of diabetes. The sensor is intended for single use and requires a p rescrip tion . The Guardian™ 4 sensor is indicated for up to seven days of continuous use.
The Guardian™ 4 sensor is not intended to be used directly to make therapy adjustments while the MiniMed™ 780G is operating in manual mode. All therapy adjustments in manual mode should be based on measurements obtained using a blood glucose meter and not on values provided by the Guardian™ 4 sensor. The Guardian™ 4 sensor has been studied and is approved for use in patients ages 7 years and older and in the arm insertion site only. Do not use the Guardian™ 4 sensor in the abdomen or other body sites including the buttocks, due to unknown or different performance that could result in hypoglycemia or hyperglycemia.
|
WARNING: Do not use the SmartGuard™ feature for people who require less than 8 units or more than 250 units of total daily insulin per day. A total daily dose of at least 8 units, but no more than 250 units, is required to operate in the SmartGuard™ feature.
|
WARNING: Do not use the MiniMed™ 780G system until appropriate training has been received from a healthcare professional. Training is essential to ensure the safe use of the MiniMed™ 780G system.
WARNING: Do not use SG values to make treatment decisions, including delivering a bolus, while the pump is in Manual Mode. When the SmartGuard™ feature is active and you are no longer in Manual Mode, the pump uses an SG value, when available, to calculate a bolus amount. However, if your symptoms do not match the SG value, use a BG meter to confirm the SG value. Failure to confirm glucose levels when your symptoms do not match the SG value can result in the infusion of too much or too little insulin, which may cause hypoglycemia or hyperglycemia.
Pump therapy is not recommended for people whose vision or hearing does not allow for the recognition of pump signals, alerts, or alarms. The safety of the MiniMed™ 780G system has not been studied in pregnant women, persons with type 2 diabetes, or in persons using other anti-hyperglycemic therapies that do not include insulin. For complete details of the system, including product and important safety information such as indications, contraindications, warnings and precautions associated with system and its components, please consult https://www.medtronicdiabetes.com/important-safety-information#minimed-780g and the appropriate user guide at https://www.medtronicdiabetes.com/download-library